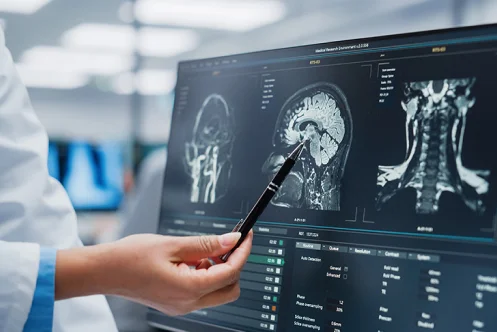
Medical Device Software Testing to Prevent FDA Recalls
We identify and address potential issues related to software functionality, interoperability, and user safety.
Our Valued Customers
Protecting Patients from Incorrect Diagnoses
Patient harm. Loss of life. Misdiagnosis. The consequences of software failures in medical devices can be severe. That is why we abide by the following pillars:
- Ensure compliance with regulatory authorities, including FDA, HIPAA, DICOM, and HL7 Standards.
- Drastically reduce release time to the market, without compromising on quality.
- Optimize UX with efficient performance testing.
Services Offered in Testing Software for Medical Devices
Handling Of Errors
We evaluate your product’s performance under critical conditions to check self-regulation and fault-tolerance features for maximum credibility.

Cyber Security
Cyber engineers at Folio3 build security into every stage of your system development lifecycle to instantly spot vulnerabilities and mitigate risks.

Usability Testing
We test your device to ensure its design and user interface satisfy the regulatory standards for safety and ease of use to improve customer satisfaction.

Load Testing
We test your medical device at high loads to ensure reliability, responsiveness, and compatibility at all times without any delay or error.

Performance Testing
We integrate performance engineering into your CI/CD pipeline so bottlenecks are detected early in the SDLC to identify any possible issues and resolve them.

Verification And Validation
Our team optimizes your V&V efforts and documents those efforts to be FDA-compliant for maximum transparency.

Your Partners in Making Medical Software Failure Proof!
Scalability and Interoperability
Scalability and Interoperability
Time and Cost Efficiency
Time and Cost Efficiency
Reliability and Performance
Reliability and Performance
Regulatory Compliance
Regulatory Compliance
Patient Safety
Patient Safety
Case Studies: How We Helped
We partnered with healthcare providers and entrepreneurs to solve global health challenges. Learn more about how Folio3 Digital Health uses its clinical brains to deliver meaningful solutions to healthcare organizations, institutions, startups, and support networks.
Healthcare Development Tech Stack
We have state-of-the-art technologies to create healthcare app solutions that effortlessly integrate with your existing infrastructure while staying ahead of industry trends.
How Our Software Development Process Work?
01
01
02
02
03
03
04
04
05
05
Client Testimonials










Kudos to your team for outstanding work in the discovery phase! Your team understood our business and workflows by asking the right questions and extracting the most useful information. Your team's ownership and results-oriented approach are commendable.
Shaukat Zakaria
CEO at HealthQuest
I’d like to thank everyone for their extra work and making the first release possible before the actual date. I'm overjoyed to see the practical aspect of our application come to life today and hoping to build great products with Folio3.
Maker Athian
Founder at iVision
We've successfully completed the first MVP and I appreciate everyone's efforts especially for the identical parts of the apps and now we have a live product on AWS through your excellent work. Thank you !
Maker Athian
Founder at iVision
I appreciate your remarkable work during the recent Laboratory service phase. Your determination, analytical thinking, and continuous efforts were crucial in overcoming challenges and achieving success. I appreciate your help as we collaborate to create fantastic items.
Maker Athian
Founder at iVision

Working with the folio3 team has been a great experience. Your dedication, creativity, and adaptability in overcoming challenges for the Moodology app project are truly commendable.
Azhar Ali
Director At AiGenics


Team Folio3 brought my dream app to reality, they explained every step to our non technical team professionally. wireframe and design processes were outstanding & they built stunning and perfectly integrated application. I think both patients and clinic will be significantly benefited.
Elizabeth Miazga
MD FRCSC at Saint Michael's Medical Center

Our Latest
Healthcare Insights
Frequently Asked Questions
If we talk about the average cost of telehealth software, then it’s safe to say that the cost of telemedicine software is around $25000 if we are talking about a basic solution with no frills.
According to the U.S. FDA, software has been defined as a medical device or SaMD (Software as a Medical Device) – a form of digital health technology aimed at enhancing medical outcomes for patients.
The FDA permits the use of software as a medical device to deliver benefits but only approves software that meets their standards. Furthermore, the FDA regulates SaMD to ensure that the software in use is both safe and relatively risk-free.
Folio3 is a software solution provider that has been delivering software solutions in all domains for over 15 years. There are multiple reasons why Folio3 is an ideal company for software development in the medical field.
The benefits include following ISO9001 standards, having medical domain experts on the team, making use of newer technology, and providing extensive documentation. These multiple benefits are critical when hiring the best software companies approved by the FDA.
- The business case is completely defined.
- The stakeholders are called for a meeting.
- Software requirements are specified.
- Domain experts analyze the domain and start to design the technical architecture.
- The Folio3 developers started to develop code for the software.
- The code is simultaneously tested for errors by the software quality engineers.
- The system is integrated and the entire system is validated as a single unit.
- Finally, the usability evaluations, clinical evaluations, and instructions are documented.
The software, functioning as a medical device, comes with a hefty price tag. According to research conducted by Starfish Medical, the total capital required to construct a Class II 510(k) FDA-approved medical device will amount to approximately $30 million. The software development and design for the medical device will constitute almost $2-5 million of this $30 million budget.
Approval costs for a Class III medical device undergoing premarket approval reach $94 million, with nearly 80% allocated to testing and revision phases outlined by the FDA. To reduce additional expenses, it is advisable to opt for the services of a company that can provide an all-in-one solution package, such as Folio3.
The medical device software development process has several stages that allow the final software for medical devices to be developed. Medical device software companies follow their development processes. Folio3 follows a process that is based on the latest international standards, their own company experience, and the rules and regulations of the FDA. The process phases are planning, code development, testing, usability design, integration with hardware, and testing the software as a single unit. The development process of Folio3 ensures that there is no lack in the software and no revisions are needed which helps cut significant costs.