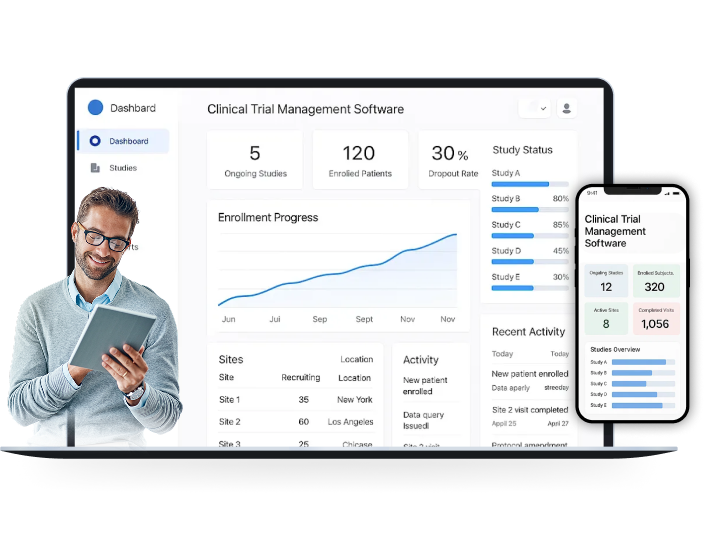
Integrated CTMS System for Clinical Research
Run smarter, faster, and fully compliant trials with our trial management solution that simplifies site operations, monitors participants in real time, and keeps all trial data secure, integrated, and transparent.

Trusted By
Healthcare Enterprises





What Our CTMS Software Delivers
Our clinical trial software helps trials run more efficiently by automating the tasks that slow teams down. Site activation moves faster, enrollment issues surface earlier, & documents stay aligned at all times. With fewer manual steps, teams manage studies with more accuracy, less risk, and a predictable path to completion.
Study & Site Management
Faster site activation, consistent submissions, and secure access to study materials for sponsors, CROs, and sites.
Participant Management
Unified participant data from EDC, e-participant report outcome, with full audit history and AI alerts for missed visits or safety risks.
Regulatory & Compliance Oversight
All regulated documents managed in one place, aligned with HIPAA & GDPR, with secure eSignatures.
Real-Time Reporting & Dashboards
Live visibility into enrollment, deviations, and milestones, with AI-driven anomaly detection and Power BI/Tableau integration.
Get Custom Add-ons
Connects with EDC, labs, wearables, billing systems, and EMR/EHR. Supports HL7 and FHIR for smooth data movement.
Investigator & Staff Portal
Shared workspace for dashboards, tasks, reminders, and training, helping teams stay aligned and follow protocols.
High-Level Platform Capabilities: What Sets Our CTMS Apart
AI-Driven Trial Oversight
Predict enrollment trends, site performance, and protocol deviations using built-in AI models. It helps optimize trial execution with intelligent, data-backed insights.

Custom Workflow & Operations Engine
Configure task flows for monitoring enrollment, deviations, timelines, and document approvals. They are aligned with SOPs using drag-and-drop automation and real-time alerts.
Reduced Administrative Burden
Our ERP solutions enhance decision-making, resource allocation, and patient care delivery with healthcare data analytics services that leverage advanced analytics, artificial intelligence (AI) predictive insights, and automated workflows.

Protocol & Regulatory Management
Keep IRB approvals, version-controlled submissions, and document lifecycles organized with a clear 21 CFR Part 11 audit trail, making it easier to stay compliant.

Real-Time Reporting & Dashboard
Track key metrics with easy-to-use interactive dashboards. These connect smoothly with Power BI, Tableau, and HL7/FHIR data outputs for streamlined insights.
Clinical Supply & Inventory Control
Forecast IP needs with AI models, automate depot-to-site shipments, and maintain a chain of custody through temperature logs and barcode scanning in clinical trial supply management software.

Participant-Centric Data Integration
Link EDC, ePRO, and eConsent for unified participant tracking in decentralized or hybrid trials. Ensure compliance and continuity across touchpoints.
Run Efficient, Compliant, and Transparent Clinical Trials
Our CTMS is built to handle the full lifecycle of clinical trials, packed with features for study management, participant tracking, reporting, and gaining real-time insights across every stage of your trials.
AI Enablement
- Predict enrollment patterns, site performance trends, and potential protocol deviations
- Automate detection of data inconsistencies with real-time query suggestions
- AI-driven alerts for missed visits, milestone delays, or participant risk indicators
- Smart recommendations for task prioritization and visit scheduling
Study & Site Management
- Real-time tracking of site activation and submissions
- Tailored workflows that match your SOPs
- Secure eTMF access through seamless API integrations

Participant Management
- Linked with EDC, ePRO, and eConsent for smooth decentralized trial execution
- 21 CFR Part 11-compliant audit trails and user-based access
- Alerts for missed visits, SAEs, or status changes in real-time

Clinical Trial Financial Management
- Automate site payments and track contract milestones
- Multi-currency support for global studies
- Plug-and-play integrations with NetSuite, SAP, and QuickBooks

Real-Time Reporting & Dashboards
- Custom dashboards to monitor enrollment, deviations, and timelines
- BI tools like Power BI and Tableau are fully integrated
- Export-ready data formats for accessibility and easy usage

Integration Hub (API-First Architecture)
- Connect EDC, eConsent, EMR/EHR, wearable devices, labs, and billing systems
- Sync protocol details, demographics, billing grids, and more
- RESTful APIs with CDISC ODM, HL7, and FHIR integration ensure secure, real-time data exchange

Investigator & Staff Portal
- Dashboards, training logs, and task tracking for PIs and CRCs
- Role-based access with automated reminders and alerts
- Reduce protocol deviations with centralized oversight

Workflow & Document Automation
- Drag-and-drop automation for alerts, approvals, and versioning
- Document locking, expiration tracking, and rollback tools
- Built-in eSignatures via DocuSign or Adobe Sign

Regulatory & Compliance Oversight
- Manage submissions and version-controlled docs in one place
- FDA 21 CFR Part 11, ICH-GCP, and HIPAA compliant
- Built-in eSignatures, timestamped actions, and role-based access

Simplified System Connections with Custom Clinical Trial Management Software
EHR & EMR Integration
Patient Demographics Interface
eConsent & EDC Integration
EHR & EMR Integration

- Sync protocol and subject data using HL7 and FHIR standards
- Automatically flag research participants through Epic integration and other EMR systems to streamline research coordination
Patient Demographics Interface
- Auto-import relevant demographic fields from EMR to CTMS to improve speed and data consistency
- Reduces staff workload and enhances accuracy in decentralized environments
eConsent & EDC Integration

- Link participant consent status and captured data with your clinical trial management software
- AI-assisted enrollment decisions and regulatory tracking across decentralized and hybrid trials
Customizable CTMS System For Your Study
Simplify your complex clinical trials with our Customizable CTMS based on your requirements. Our clinical trial management solution has enhanced integration capabilities, like EDC, RTSM, to simplify research management.
-
EDC – Electronic Data Capture
Build, launch, and manage complex studies with our flexible platform that makes clean data easier to capture and manage. -
ePRO – Electronic Patient-Reported Outcomes
Let participants report from anywhere with a centralized system that keeps participants engaged and data collection on track. -
eConsent – Electronic Informed Consent
Streamline informed consent with digital forms and clear, trackable workflows that simplify compliance and empower participants. -
RTSM – Randomization and Trial Supply Management
Manage randomization and track trial supply in one seamless system, tailored to your protocol and designed to keep sites moving.
Get in Touch
© 2025 Folio3 Software Inc., All Rights Reserved.

